Heart Failure with Preserved Ejection Fraction (HFpEF)
Definition
HFpEF is defined by the presence of HF symptoms ± signs, LVEF ≥50%, and objective evidence of cardiac structural and/or functional abnormalities consistent with LV diastolic dysfunction/raised LV filling pressures, including raised natriuretic peptides. The greater the number of abnormalities present, the higher the likelihood of HFpEF. (sources/HF-ESC-2021, rating: very high)
Epidemiology
- HFpEF accounts for ~16% of outpatient HF in ESC Long-Term Registry; prevalence increases with age. (sources/HF-ESC-2021, rating: very high)
- Comorbidities (hypertension, obesity, T2DM, CKD, AF) are highly prevalent in HFpEF and drive pathophysiology. (sources/HF-ESC-2021, rating: very high)
- Obesity and HFpEF: HFpEF is more strongly associated with obesity than HFrEF. Overweight → 38% higher HFpEF risk; class 1 obesity → 56% higher HFpEF risk, independently of other CVD risk factors (Pandey et al., pooled analysis of 3 longitudinal studies). (sources/obesity-cv-aha-2021, rating: very high)
- Normal natriuretic peptide levels do not exclude HFpEF in obese patients — adipose tissue clears BNP, reducing measured levels even in the presence of elevated filling pressures. (sources/obesity-cv-aha-2021, rating: very high)
Pathophysiology
Comorbidity-Driven Mechanisms
- Systemic comorbidities (hypertension, obesity, T2DM, CKD, AF) promote coronary microvascular inflammation, cardiomyocyte hypertrophy, and interstitial fibrosis — the dominant pathophysiological substrate in HFpEF. (sources/HF-ESC-2021, rating: very high)
- Increased cardiomyocyte passive stiffness is a hallmark of HFpEF, arising from both extracellular matrix changes and intrinsic sarcomeric protein modifications.
Obesity-HFpEF Phenotype
- Obesity produces a distinct HFpEF phenotype compared with non-obese HFpEF: greater concentric LV remodeling, RV dilatation and dysfunction, pericardial restraint, ventricular interdependence, and significantly lower exercise capacity. These features are driven by greater epicardial fat thickness and volume — the first study to formally characterize this as a distinct obesity-HFpEF pathophysiology. (sources/obesity-cv-aha-2021, rating: very high)
- Visceral adiposity → higher blood volume and cardiac output → LVH and LV diastolic dysfunction → HFpEF. Obesity also causes myocardial fat accumulation and fibrosis through epicardial adipose tissue paracrine signaling. (sources/obesity-cv-aha-2021, rating: very high)
- The H2FPEF score includes Obesity (BMI >30) as the highest-weighted component (2 of 9 points), reflecting the dominant role of obesity in HFpEF clinical probability. (sources/HF-AHA-2022, rating: very high)
- See entities/Obesity and concepts/Visceral-Adiposity for full mechanistic detail.
Titin Stiffness Mechanisms
- Titin contributes substantially to cardiomyocyte passive stiffness through multiple post-translational mechanisms. (sources/TTN-CVResearch-2022, rating: high)
- Isoform shift: Increased N2BA:N2B ratio observed in some HFpEF patients → lower titin-based stiffness as an apparent compensatory mechanism. The direction and functional significance remain inconsistent across studies. (sources/TTN-CVResearch-2022, rating: high)
- Phosphorylation: N2Bus hypo-phosphorylation (reduces extensibility) + PEVK hyper-phosphorylation (increases stiffness) → combined increase in passive stiffness. Both PKA/PKG-mediated N2Bus phosphorylation and the cGMP-PKG pathway are impaired in HFpEF. (sources/TTN-CVResearch-2022, rating: high)
- Acetylation: Reduced SIRT1 activity in HFpEF → increased titin acetylation → increased stiffness. NAD⁺/NAM treatment restores SIRT1 → deacetylation of titin spring → reduced stiffness in animal models. (sources/TTN-CVResearch-2022, rating: high)
- Failed clinical trials targeting titin phosphorylation via cGMP-PKG:
- RELAX (sildenafil/PDE5A inhibitor): no improvement in diastolic function or VO2 in HFpEF. (sources/TTN-CVResearch-2022, rating: high); (sources/HF-AHA-2022, rating: very high)
- VITALITY and SOCRATES (sGC stimulators): no improvement in HFpEF despite pre-clinical evidence of N2Bus phosphorylation increase and reduced cardiomyocyte stiffness. (sources/TTN-CVResearch-2022, rating: high)
- See concepts/Titin-PTMs and concepts/Titin-Isoform-Switch for full mechanistic detail.
Genetic and Infiltrative Causes
- Anderson–Fabry Disease (AFD): 40% of AFD patients meet ESC HF criteria; 91% of those have HFpEF (rather than HFrEF). Subclinical diastolic dysfunction may precede LVH — symptoms can appear before significant wall thickening. (sources/fabry-ehj-2024, rating: high)
- ATTR cardiac amyloidosis (ATTR-CA): Prevalence in HFpEF estimated at 5–13% (limited autopsy/nuclear screening data); one prospective biopsy study found 14% of HFpEF had cardiac amyloidosis, majority TTR type. (sources/echo-hfpef-ase-2025, rating: very high)
- See entities/Fabry-Disease, concepts/Fabry-Cardiomyopathy, and entities/ATTR-Amyloidosis for full detail.
Atrial Myopathy in HFpEF
- In HFpEF, mechanical atrial dysfunction (impaired LASr, elevated LAVi) is an independent and better predictor than AF for adverse clinical outcomes including morbidity (HF) and cardiovascular mortality. AF alone is insufficient to capture the full atrial risk in this population. (sources/atrial-cmp-esc-2025, rating: high)
- LASr correlates with histological atrial fibrosis in advanced HF patients, making echocardiographic LASr a surrogate measure of atrial fibrosis — relevant for both diagnosis and prognostication in HFpEF. (sources/atrial-cmp-esc-2025, rating: high)
- A subset of HFpEF is characterized by 'disproportionate LA myopathy' — LA dysfunction more pronounced than expected from LV dysfunction alone. Right heart dysfunction, enhanced left-to-right atrial interaction, and pericardial constraint may contribute. (sources/atrial-cmp-esc-2025, rating: high)
- The obesity-HFpEF phenotype features more atrial epicardial adipose tissue when AF co-occurs, contributing to atrial remodelling via paracrine fibrotic/inflammatory signalling. (sources/atrial-cmp-esc-2025, rating: high)
- This disproportionate LA myopathy may represent a distinct HFpEF phenotype or an early-stage manifestation of atrial failure — the distinction requires further study.
- See concepts/Atrial-Cardiomyopathy and concepts/Atrial-Failure for the full AtCM diagnostic framework.
Clonal Hematopoiesis (TET2)
- TET2-CH is 2.4-fold enriched in HFpEF — a gene-specific association not seen with the overall HFrEF/HFpEF classification, identifying CH as a potential contributor to HFpEF pathophysiology specifically. (sources/ch-aha-2026, rating: very high)
- Mechanism: TET2 loss-of-function → NLRP3 inflammasome overactivation → increased IL-1β secretion; elevated circulating IL-1β is unique to TET2-CH among all CH variants and is the likely mediator of the cardiac inflammatory phenotype. (sources/ch-aha-2026)
- CANTOS post-hoc: canakinumab (anti-IL-1β) reduced ischemic events far more in TET2-CH carriers — mechanistic relevance for HFpEF inflammatory phenotype under investigation. (sources/ch-aha-2026)
- See concepts/Clonal-Hematopoiesis for full gene-specific mechanisms, emerging therapeutic strategies, and the broader CH–HF risk association (25% overall HF risk increase in CH carriers).
Diagnosis
LVEF-Based Phenotype Classification
- HFpEF is one of three LVEF-based HF phenotypes: HFrEF (LVEF ≤40%), HFmrEF (LVEF 41–49%), HFpEF (LVEF ≥50%). (sources/HF-ESC-2021, rating: very high)
- The 2023 ESC task force considered renaming HFpEF to HFnEF (normal EF) but retained current terminology, deferring to the next full guideline update. (sources/HF-update-ESC-2023, rating: very high)
- In clinical trials (EMPEROR-Preserved, DELIVER): NT-proBNP thresholds used were >300 pg/mL for sinus rhythm and >900 pg/mL for AF. These thresholds were not mandated in the 2023 recommendation. (sources/HF-update-ESC-2023, rating: very high)
Guideline Definitions: ESC vs ACC/AHA
- ACC/AHA: LVEF ≥50% + symptoms/signs + evidence of diastolic dysfunction. (sources/echo-hfpef-ase-2025, rating: very high)
- ESC: LVEF ≥50% + symptoms/signs + elevated natriuretic peptides + at least one of:
- Structural: LAVi >34 mL/m² or LVMi ≥115/95 g/m² (men/women)
- Functional: E/e' ≥13 or mean e' <9 cm/s (sources/echo-hfpef-ase-2025, rating: very high)
- Up to 30% of HFpEF patients can have normal NP levels (particularly obese patients) — normal NP does not exclude HFpEF. (sources/echo-hfpef-ase-2025, rating: very high)
HFpEF Prediction Scores
- H2FPEF Score (0–9): Discriminates HFpEF from noncardiac dyspnoea. Components: Heavy (obesity, BMI >30; 2 pts), Hypertensive (≥2 antihypertensives; 1 pt), Atrial fibrillation (3 pts), Filling pressure (E/e' >9; 1 pt), Elderly (age >60 years; 1 pt), Pulmonary hypertension (PASP >35 mmHg; 1 pt). Each 1-unit increase doubles odds of HFpEF (OR 1.98; c-statistic 0.841). (sources/HF-AHA-2022, rating: very high)
- Score <2 = low likelihood; 2–5 = intermediate (40–80%); ≥6 = high likelihood
- Intermediate scores: refer for exercise echocardiography or invasive hemodynamics
- HFA-PEFF Algorithm (ESC): Stepwise pretest probability → comprehensive echo + NP score → exercise/invasive testing if intermediate. (sources/echo-hfpef-ase-2025, rating: very high)
- The 2025 ASE guideline formally incorporates both H2FPEF and HFA-PEFF into HFpEF diagnostic workup for patients with uncertain probability. (sources/echo-hfpef-ase-2025, rating: very high)
ASE 2025 — Stepwise Echocardiographic Diagnosis Algorithm
Comprehensive stepwise algorithm from the 2025 ASE guideline update: (sources/echo-hfpef-ase-2025, rating: very high)
- History, physical, CXR, labs, natriuretic peptides → exclude non-cardiac dyspnea
- Comprehensive echo → exclude: significant valvular disease (MS/primary MR/AS/AR/TR), significant CAD, non-cardiac PH, cardiac amyloidosis, HCM, pericardial constriction
- Apply LAP estimation algorithm (e', E/e', TR velocity/PASP as primary variables)
- If LAP elevated → HFpEF confirmed
- If LAP normal → diastolic exercise echo (or RHC with/without exercise)
- If exercise echo positive → HFpEF confirmed; if negative → non-cardiac dyspnea; if inconclusive → RHC
RHC gold standard: Rest mean PCWP >15 mmHg or exercise mean PCWP ≥25 mmHg (end-expiration) confirms HFpEF. (sources/echo-hfpef-ase-2025, rating: very high)
Definition of LV Diastolic Dysfunction (ASE 2025 Two-Step Algorithm)
A two-step approach is used to define the presence of LV diastolic dysfunction. (sources/echo-hfpef-ase-2025, rating: very high)
Step 1 — Assess impaired LV relaxation (e' velocity):
- Reduced e' defined as: septal e' ≤6 cm/s OR lateral e' ≤7 cm/s OR average e' ≤6.5 cm/s
Step 2 — Assess LA/LV remodeling and elevated LAP (≥1 marker required):
| Marker | Abnormal Threshold |
|---|---|
| Average E/e' | >14 |
| LA reservoir strain (LARS) | ≤18% |
| E/A ratio | ≤0.8 (age-adjusted) OR ≥2 |
| LAVi | >34 mL/m² |
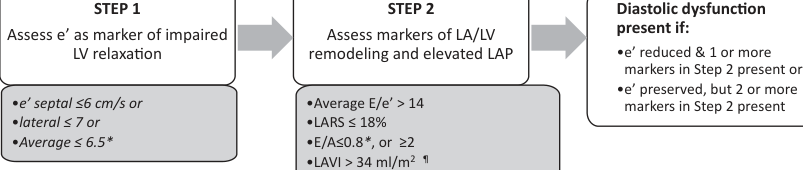
Diagnostic rule:
- Diastolic dysfunction present if: reduced e' + ≥1 Step 2 marker, OR normal e' + ≥2 Step 2 markers
- Additional supportive finding: LV mass index >115 g/m² (men) or >95 g/m² (women), excluding athletes
Key change from 2016 guideline: LAVi demoted from primary to secondary variable in the LAP estimation algorithm — poor correlation with acute LAP changes and multiple non-diastolic causes of LA enlargement (AF, MR, high-output states). LARS (≤18%) elevated to primary parameter with high specificity for elevated LAP. (sources/echo-hfpef-ase-2025, rating: very high)
Echocardiographic HFpEF Features
- Typical features: LV hypertrophy, concentric remodeling, LA enlargement, diastolic dysfunction (Grade 2 or 3), reduced LV GLS, reduced LARS — but none are individually diagnostic. (sources/echo-hfpef-ase-2025, rating: very high)
- See concepts/LV-Diastolic-Function for the full LAP estimation algorithm and diastolic dysfunction grading.
Diastolic Exercise Echocardiography
- Up to 50% of HFpEF patients have normal resting LAP that rises only with exercise — exercise echo is essential in this phenotype. (sources/echo-hfpef-ase-2025, rating: very high)
- Positive (definite abnormal): Average E/e' ≥14 (or septal ≥15) AND peak TR velocity >3.2 m/s during exercise. (sources/echo-hfpef-ase-2025, rating: very high)
- Likely HFpEF: E/e' >14 + TR velocity >2.8 but <3.2 m/s. (sources/echo-hfpef-ase-2025, rating: very high)
- Normal: Average E/e' <10 AND TR velocity <2.8 m/s. (sources/echo-hfpef-ase-2025, rating: very high)
Management
Historical Pharmacotherapy (Pre-SGLT2i Era)
- As of 2021 ESC guidelines: no pharmacotherapy shown to reduce mortality in HFpEF. ACE-I, ARB, MRA, ARNI all failed to meet primary endpoints. (sources/HF-ESC-2021, rating: very high)
- Treatment focused on managing aetiologies and comorbidities, and symptom relief with diuretics. (sources/HF-ESC-2021, rating: very high)
ESC 2023 — SGLT2 Inhibitors (Class I, Level A)
- EMPEROR-Preserved (empagliflozin, n=5988, LVEF >40%, median follow-up 26.2 months): Composite of CV death or HF hospitalization reduced (HR 0.79, 95% CI 0.69–0.90; P<0.001). Effect driven by reduced HF hospitalizations; no significant reduction in CV death. (sources/HF-update-ESC-2023, rating: very high)
- DELIVER (dapagliflozin, n=6263, LVEF >40%, including improved LVEF): Primary endpoint of CV death or worsening HF reduced (HR 0.82, 95% CI 0.73–0.92; P<0.001). Benefit from worsening HF component; no reduction in CV death. Consistent benefit across full LVEF range and in the improved-LVEF subgroup. (sources/HF-update-ESC-2023, rating: very high)
- Pooled meta-analysis: 20% reduction in CV death or first HF hospitalization (HR 0.80; P<0.001); 26% reduction in HF hospitalization (HR 0.74; P<0.001). CV death not significantly reduced (HR 0.88; P=0.052). (sources/HF-update-ESC-2023, rating: very high)
- 2023 Recommendation: SGLT2 inhibitor (dapagliflozin or empagliflozin) Class I, Level A to reduce risk of HF hospitalization or CV death. Benefit independent of T2DM status. (sources/HF-update-ESC-2023, rating: very high)
- ESC 2023 Algorithm — Class I: Diuretics for fluid retention; SGLT2 inhibitor; treatment of aetiology, CV and non-CV comorbidities. Management remains heavily comorbidity-directed (AF, hypertension, diabetes, obesity, CKD, sleep apnoea). (sources/HF-update-ESC-2023, rating: very high)
SGLT2 Inhibitors in HFmrEF
- HFmrEF (LVEF 41–49%): SGLT2 inhibitors upgraded to Class I, Level A (from Class IIb/no recommendation in 2021). Evidence base is the same two trials (EMPEROR-Preserved and DELIVER enrolled LVEF >40%). (sources/HF-update-ESC-2023, rating: very high)
- In HFmrEF, other agents (ACE-I/ARNI/ARB, beta-blocker, MRA) remain Class IIb based on subgroup analyses; diuretics remain Class I for fluid retention. (sources/HF-update-ESC-2023, rating: very high)
- SGLT2 inhibitors should be considered in AFD-related HFpEF per ESC Class I recommendation; they may also confer additional renal benefit in AFD nephropathy. (sources/fabry-ehj-2024, rating: high)
- Cardiac myosin inhibitors (mavacamten) must not be used in AFD-HFpEF — approved only for sarcomeric obstructive HCM. (sources/fabry-ehj-2024, rating: high)
AHA 2022 — Pharmacotherapy (COR Levels)
- BP control: COR 1, LOE C-LD — titrate to published BP targets. (sources/HF-AHA-2022, rating: very high)
- SGLT2i: COR 2a, LOE B-R — beneficial for HF hospitalizations and CV mortality (EMPEROR-Preserved). (sources/HF-AHA-2022, rating: very high)
- AF management: COR 2a, LOE C-EO — useful for symptom improvement. (sources/HF-AHA-2022, rating: very high)
- MRA: COR 2b, LOE B-R — particularly at lower LVEF spectrum (TOPCAT data). (sources/HF-AHA-2022, rating: very high)
- ARB: COR 2b, LOE B-R — particularly at lower LVEF (CHARM-Preserved data). (sources/HF-AHA-2022, rating: very high)
- ARNi: COR 2b, LOE B-R — particularly at lower LVEF (PARAGON-HF subgroup). (sources/HF-AHA-2022, rating: very high)
- Nitrates and PDE5 inhibitors: COR 3 No Benefit, LOE B-R — routine use ineffective. NEAT-HFpEF: no improvement in activity tolerance with isosorbide mononitrate. RELAX: no improvement in VO2 or exercise tolerance with sildenafil. (sources/HF-AHA-2022, rating: very high)
Guideline Discordance: AHA 2022 vs ESC 2023 on SGLT2i
- AHA 2022: SGLT2i COR 2a — based solely on EMPEROR-Preserved. Published May 2022, before DELIVER (August 2022). (sources/HF-AHA-2022, rating: very high)
- ESC 2023: SGLT2i Class I, Level A — incorporates both EMPEROR-Preserved and DELIVER plus pooled meta-analysis. (sources/HF-update-ESC-2023, rating: very high)
- Both guidelines agree SGLT2i is beneficial in HFpEF; the difference in classification strength reflects publication timing, not disagreement on efficacy.
Contradictions / Open Questions
- Mechanical atrial dysfunction vs. AF in HFpEF: ESC/HFA 2025 AtCM consensus shows mechanical atrial dysfunction is a better predictor than AF in HFpEF — yet HFpEF management frameworks (AHA/ESC) continue to rely heavily on AF detection as a primary driver of management decisions (NT-proBNP thresholds, OAC, rate/rhythm control). This tension suggests atrial structural markers may need formal incorporation into HFpEF diagnostic and management algorithms. (sources/atrial-cmp-esc-2025, rating: high; sources/echo-hfpef-ase-2025, rating: very high)
- SGLT2i reduces HF hospitalizations but not CV death in HFpEF: Both EMPEROR-Preserved and DELIVER showed no significant reduction in CV death individually; the pooled meta-analysis also showed a non-significant trend (HR 0.88, P=0.052). The Class I, Level A recommendation rests on the composite endpoint driven by hospitalizations. (sources/HF-update-ESC-2023, rating: very high)
- Improved EF phenotype (HFrecEF): DELIVER included patients with LVEF previously ≤40% that had improved to >40%. Dapagliflozin showed consistent benefit in this subgroup — suggesting SGLT2i may benefit "recovered EF" HF, but guideline classification of this subgroup remains undefined. (sources/HF-update-ESC-2023, rating: very high)
- NT-proBNP thresholds not mandated in recommendation: Trial entry required elevated natriuretic peptides; the 2023 task force chose not to specify thresholds in the recommendation text, creating uncertainty about treating HFpEF with low natriuretic peptides. (sources/HF-update-ESC-2023, rating: very high)
- Titin isoform switch direction in HFpEF is inconsistent: Some studies show increased N2BA:N2B ratio (lower stiffness), others do not — the role of isoform switching as compensatory vs. pathological in HFpEF remains unresolved. (sources/TTN-CVResearch-2022, rating: high)
- cGMP-PKG pathway as therapeutic target has failed in clinical trials: Despite strong pre-clinical mechanistic rationale (RELAX, VITALITY, SOCRATES all negative), suggesting the titin phosphorylation model may be insufficient or not translatable in humans. (sources/TTN-CVResearch-2022, rating: high)
Connections
- Related to concepts/Atrial-Cardiomyopathy — mechanical atrial dysfunction independent predictor in HFpEF; disproportionate LA myopathy phenotype
- Related to concepts/Atrial-Failure — atrial failure and HFpEF phenotypes overlap
- Related to entities/Heart-Failure
- Related to concepts/LV-Diastolic-Function — echo-based HFpEF diagnosis algorithm
- Related to entities/Atrial-Fibrillation — elevated NT-proBNP thresholds differ in AF patients
- Related to entities/Fabry-Disease — AFD as genetic cause of HFpEF
- Related to concepts/Fabry-Cardiomyopathy
- Related to entities/ATTR-Amyloidosis — infiltrative cause of HFpEF
- Related to concepts/Titin-PTMs
- Related to concepts/Titin-Isoform-Switch
- Related to sources/HF-ESC-2021
- Related to sources/HF-update-ESC-2023
- Related to sources/HF-AHA-2022
- Related to sources/echo-hfpef-ase-2025
- Related to sources/TTN-CVResearch-2022
- Related to sources/fabry-ehj-2024
- Related to entities/Obesity — obesity-HFpEF distinct phenotype
- Related to concepts/Visceral-Adiposity — epicardial fat and pericardial restraint in obesity-HFpEF
- Related to concepts/Obesity-Paradox — less consistent in HFpEF than HFrEF
- Related to sources/obesity-cv-aha-2021
- Related to sources/atrial-cmp-esc-2025
- Related to sources/ch-aha-2026
- Related to concepts/Clonal-Hematopoiesis — TET2-CH 2.4× enriched in HFpEF; NLRP3/IL-1β mechanism